Voiceover Our goal is to calculate the equilibrium constant K for this reaction, so for this reaction right here. Now we're gonna use the standard reduction potentials to do so. So in the previous video, we talked about the relationship between the equilibrium constant K and the standard cell potential. Sep 24, 2016 Find Kb for ammonia: This would simply be the given Keq for the reaction. Kb is just the Keq of a specific type of reaction between a base and water. Ammonia is a weak base. Find Ka of conjugate acid. This is would equal the Keq of the reverse reaction. The Keq of the reverse reaction is the reciprocal of the the forward reaction. Calculate the concentrations and substitute them into an expression and compute the 'experimental concentration quotient' or 'Q'. If Q is equal to K eq (50 in this case) then the system is already at equilibrium and stable. If Q K eq then the product is too high and must decrease. Therefore the reverse reaction is favoured. Calculate Keq for 2 moles of electron(s) in each half-reaction and a standard potential of 0.55 V. Standard Electrode Potential: The measure of the individual potential for a reversible electrode. Step 9: Calculate the equilibrium constant for each of the test solution, average the Keq for each test solution to find the average Keq of the iron (III) thiocyanate solution and average deviation of the average Keq value. This is done by using the results obtained in Step 6 (INITIAL concentrations of Fe3+ and SCN–) and Step 8 (EQUILIBRIUM.
How To Calculate Keq
Calculating Equilibrium Constants
We need to know two things in order to calculate the numeric value of the equilibrium constant:
- the balanced equation for the reaction system, including the physical states of each species. From this the equilibrium expression for calculating Kc or Kp is derived.
- the equilibrium concentrations or pressures of each species that occurs in the equilibrium expression, or enough information to determine them. These values are substitued into the equilibrium expression and the value of the equilibrium constant is then calculated.
- Write the equilibrium expression for the reaction.
- Determine the molar concentrations or partial pressures of each species involved.
- Subsititute into the equilibrium expression and solve for K.
CO + H2O' height =34 src='EquilibriumArt/CalcKexample1.gif' width=288 NOSAVE>
- Write the equlibrium expression for the reaction system.
- Since Kc is being determined, check to see if the given equilibrium amounts are expressed in moles per liter (molarity). In this example they are not; conversion of each is requried.
[CO2] = 0.1908 mol CO2/2.00 L = 0.0954 M
[H2] = 0.0454 M
[CO] = 0.0046 M
[H2O] = 0.0046 M - Substitute each concentration into the equilibrium expression and calculate the value of the equilibrium constant.
Calculating K from Initial Amounts and One Known Equilibrium Amount
- Write the equilibrium expression for the reaction.
- Determine the molar concentrations or partial pressures of each species involved.
- Determine all equilibrium concentrations or partial pressures using an ICE chart.
- Substitute into the equilibrium expression and solve for K
N2 + 2 H2O' height=32 src='EquilibriumArt/CalcKexample2.gif' width=326 >
- Write the equilibrium expression for the reaction.
- Check to see if the amounts are expressed in moles per liter (molarity) since Kc is being . In this example they are.
- Create an ICE chart that expresses the initial concentration, the change in concentration, and the equilibrium concentration for each species in the reaction. From the chart you can determine the changes in the concentrations of each species and the equilibrium concentrations. From the example, we start with the folowing information.
How To Calculate Keq From Delta G
| Initial Concentration (M) | ||||
| Change in Concentration (M) | - 2 x | - 2 x | + x | + 2 x |
| Equilibrium Concentration (M) |
The change in concentration of the NO was (0.062 M - 0.100M) = - 0.038 M. Thus -2 x = - 0.038 and x = 0.019. Note: the negative sign indicates a decreasing concentration, not a negative concentration. The changes in the other species must agree with the stoichiometry dictated by the balance equation. The hydrogen will also change by - 0.038 M, while the nitrogen will increase by + 0.019 M and the water will increase by + 0.038 M. From these changes we can complete the chart to find the equilibrium concentrations for each species.
| Initial Concentration (M) | ||||
| Change in Concentration (M) | ||||
| Equilibrium Concentration (M) |
- Substitute the equilibrium concentrations into the equilibrium expression and solve for Kc.
Calculating K from Known Initial Amounts and the Known Change in Amount of One of the Species
- Write the equilibrium expression for the reaction.
- Determine the molar concentrations or partial pressures of each species involved.
- Determine all equilibrium concentrations or partial pressures using an ICE chart.
- Substitute into the equilibrium expression and solve for K.
Free solo 123movies. 2 NO2' height =29 src='EquilibriumArt/CalcKexample3.gif' width=181 NOSAVE>

- Write the equilibrium expression to find Kp.
- Check to see that the given amounts are measured in appropriate pressure units since Kp is to be . In this example they are (atmospheres).
- Create an ICE chart and calculate the changes in pressure and equilibrium pressures for each species.
| Initial Pressure (atm) | ||
| Change in Pressure (atm) | ||
| Equilibrium Pressure (atm) |
- Substitute the equilibrium pressures into the expression for Kp and solve for Kp.
Great Thanks to Tina Nye (A-5; 05-06) for much work on graphics: making a rough idea reality!
Chemical equilibrium
A system in equilibrium is like our ants up there! As long as the ants work at the same speed, the piles of sand remain in equilibrium. Neither gets bigger or smaller (It is important to note however that the piles are not the same size!). The work that each ant does exactly offsets what the other is doing. Products are being stacked up and the same rate that they are being taken away. Reactants are being stacked up at exactly the same rate that they are being taken away. If this were a chemical reaction, reactants would be colliding to make product at the same rate that products would be colliding with each other to make fresh reactant again! The reaction never really stops, but the forward reaction and reverse reaction proceed and the same rate, so we don't see a change in the size of the piles! Nifty Huh!!!!?
Chemical equilibrium is reached when the rates for the forward and reverse chemical reactions are equal for a chemical system. Or you could say when products are being made as fast as they are breaking down to form reactants again. The concentrations of products and reactants are generally NOT EQUAL.
To know what the concentrations are, you use the Equilibrium Constant expression (Keq).
Keq = [products]/[reactants]. Its a ratio : ) So Keq > 1 favors products, Keq < 1 favor reactants.
For a general equation like:
wA + xB <> yC + zD
Remember, the square brackets mean concentration, in molarity, if it is given in some other unit you must calculate molarity. Coefficients in the chemical equation become exponents in the Keq expression. Leave out any solids or pure liquids since they have undefined molarities ; ).
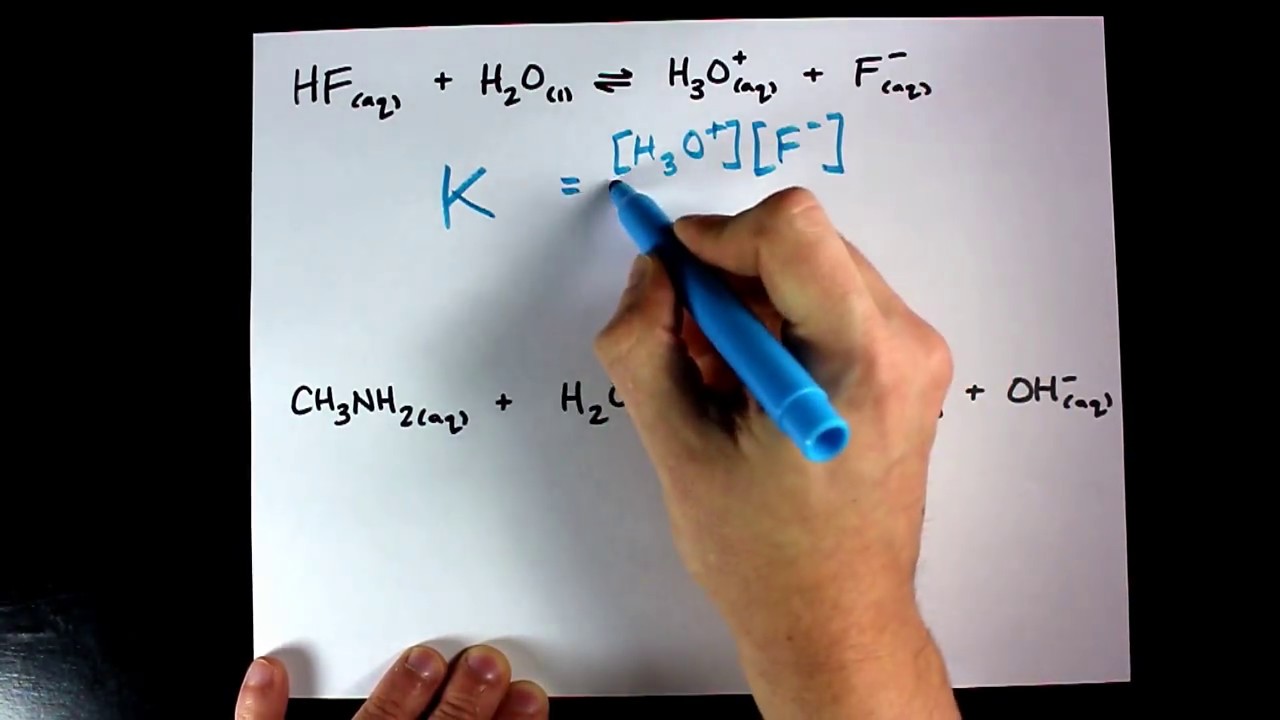
Keq= If you spend time with the following notes, you'll master this topic in no time.
Predicting final concentrations and cheating while were doing it.
